Your Courage.
Our Expertise.
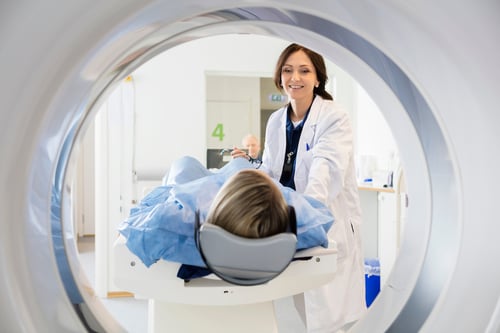
Trust your care to Colorado's leading cancer specialists.
When you’re diagnosed with cancer, the path in front of you is crucially dependent on who is behind you.

At Rocky Mountain Cancer Centers you get an entire team of doctors, nurses, and treatment professionals who have one thing in common: a relentless dedication to your health.
We fight alongside you, providing personalized, world-class care with the utmost sensitivity and kindness. Here, you can be confident you’ll get state-of-the-art treatment and the absolute best care possible.
Put Rocky Mountain Cancer Centers on your team.

![]()
Rocky Mountain Cancer Centers is the largest multidisciplinary practice in Colorado dedicated solely to providing care for patients with cancer and diseases of the blood.
Specializing in the Treatment of Nearly All Types of Adult Cancer and Blood Disorders
We Never Stop Working for You
Our team of the most dedicated specialists are driven to go above and beyond to help make you well. We listen, learn, and provide coordinated, personalized care that treats the whole you. This strategic, compassionate approach to cancer care promotes optimal patient outcomes.
![]()
9 out of 10 patients would recommend Rocky Mountain Cancer Centers
to their friends and family, according to Net Promoter Score survey results.
We Never Stop Working for You
Through our membership in The US Oncology Network, we are able to offer state-of-the-art, nationally vetted treatments and access to more clinical trials than any other cancer facility in Colorado.
![]()
Rocky Mountain Cancer Centers has played a role in more than 100 FDA-approved cancer therapies.
Cancer Education Center
Cancer comes with a lot of questions. The Rocky Mountain Cancer Centers team is dedicated to helping patients through their cancer journeys.
Our Education Center provides answers and tips you might not find elsewhere.

Caring For and Supporting Those Who Are Caring for You
A cancer diagnosis and the subsequent treatment also impact caregivers. They too feel stress and uncertainty and can find it difficult to maintain emotional balance. We want everyone on your team to stay healthy – physically, emotionally, and mentally – so they can be there with the strength, love, and care that you need.

Answering Your Financial Questions with Our Financial Counselors
One of our patient financial counselors will become an important member of your team. This valuable ally will help you understand the financial aspects of your treatment, so you can set your mind at ease and focus on getting well. At RMCC, we want to make sure your cancer treatment is all about you.
Here's What Our Patients Say

In 2017, Lakewood, Colorado resident Lois Brady wasn’t feeling very well, so she scheduled an appointment with her primary care provider. What she thought would be a routine visit marked the start of her breast cancer treatment journey.
Lois Brady
Breast Cancer Survivor
Tad Chance is a family guy who loves sports and values quality time with loved ones. He has a big, almost Brady Bunch-style family—a wife, three sons, and two stepdaughters. He loves spending time with them and watching his kids play sports, including his son who plays in an intramural league at Colorado State University, or taking his family to see professional games.
Tad Chance
Multiple Myeloma Survivor
Stan Pentecost went in for a routine annual physical in 2020 when doctors found a prostate tumor the size of a plum. Without any signs or symptoms, Stan was diagnosed with stage 3 prostate cancer.
Stan Pentecost
Prostate Cancer Survivor
One Sunday afternoon in March 2021, Brandon Martin, 61, was hanging out in his Thornton apartment when he doubled over with pain.
“All of a sudden I had a searing, intense pain in my abdomen,” Brandon said. “Then I collapsed.”
Brandon Martin
Colorectal Cancer Survivor
When Dominique Journet was told she had a tumor after a colonoscopy, she was convinced it couldn’t be cancer. She was otherwise healthy with no known risk factors. However, after a biopsy, she was diagnosed with stage 3 colon cancer.
Dominique Journet
Colorectal Cancer SurvivorOur patients are at the heart of everything we do and each story is unique. Help others navigate through their health journey by sharing your experience with us to be published on our blog.
Share Your Story >
Cancer Screening: The Next Life Saved Could Be Yours
Get to know your risk of developing cancer and when to be screened. The earlier cancer is found, the easier it is to treat. Screening saves lives! Get familiar with what's right for you.
Living Life After Cancer
There’s more to being a cancer survivor than simply completing cancer treatment. Even after treatment, we’re still on your team, offering support as you start to think about what comes next.