Non-Hodgkin Lymphoma Treatment Options
Different types of doctors often work together to create a patient’s overall treatment plan that combines different types of treatments. You can expect some form of chemotherapy, radiation therapy, immunotherapy, targeted therapy or a combination of treatments. Bone or blood marrow, as well as stem cell transplantation, may also sometimes be done under special circumstances.

If you’ve been diagnosed with non-hodgkin lymphoma, your Rocky Mountain Cancer Centers’ (RMCC) oncologist or hematologist will discuss your treatment choices and expected results with you. Once you understand what to expect, your lymphoma specialist, as well as the rest of the cancer care team, will work together to develop a treatment plan that meets your needs.
There are several factors that will affect your choice of treatment. These can include:
- The type of lymphoma you have
- Its stage (where the lymphoma is found)
- How quickly the cancer is growing (whether it is indolent or aggressive lymphoma)
- Your age
- Whether you have other health problems
Watchful Waiting
Depending on your particular situation, you may choose to put off having cancer treatment until you have symptoms. This is known as watchful waiting. If you have indolent (slow-growing) lymphoma, this might be something your RMCC oncologist recommends. People with indolent lymphoma may not have problems that require cancer treatment for a long time. Sometimes the tumor may even shrink for a while without therapy. By putting off treatment, the side effects of chemotherapy or radiation therapy can be avoided.
If you and your doctor agree that watchful waiting is a good idea, it is likely you’ll be checked regularly (every 3 months), only receiving treatment if symptoms occur or get worse. Some people do not choose watchful waiting because they don’t want to worry about having cancer that is not treated. If this is something you choose, but later become worried, please discuss your feelings with your doctor.
Find a Lymphoma Specialist Near You
Advances in Non-Hodgkin Treatment
Chemotherapy for Lymphoma
Chemotherapy for non-hodgkin lymphoma uses drugs to kill lymphoma cells. Because the drugs travel through the bloodstream, it is called systemic therapy. The drugs can reach lymphoma cells in almost all parts of the body.
There are different ways you may receive chemotherapy, which can include by mouth, through a vein, or in the space around the spinal cord. Treatment is usually in an outpatient part of the hospital, at the doctor’s office, or at home. Some people need to stay in the hospital during treatment.
Chemotherapy is given in cycles, meaning you have a treatment period followed by a rest period. The length of the rest period and the number of treatment cycles depend on the stage of your disease and on the anticancer drugs used.
If you have lymphoma in the stomach caused by H. pylori infection, your doctor may treat this lymphoma with antibiotics. After the drug cures the infection, the lymphoma also may go away.
Immunotherapy for Lymphoma
Immunotherapy, also called biologic therapy, is a treatment that uses the patient’s immune system to fight cancer. Substances made by the body or made in a laboratory are used to boost, direct, or restore the body’s natural defenses against cancer. This cancer treatment is a type of biologic therapy.
- Tafasitamab plus lenalidomide for patients who had received prior treatment, had relapsed, and had received prior autologous stem cell transplant or were not candidates for stem cell transplant.
- Tazemetostat helps many patients with relapsed follicular non-Hodgkin lymphoma with few side effects.
- Selinexor for patients who have undergone at least two lines of systemic therapy for non-Hodgkin lymphoma.
- CAR T-cell therapy uses the patient's T cells (a type of immune system cell) are changed so they will attack certain proteins on the surface of cancer cells. T cells are taken from the patient and special receptors are added to their surface in the laboratory. The changed cells are called chimeric antigen receptor (CAR) T cells. The CAR T cells are grown in the laboratory and given to the patient by infusion. The CAR T cells multiply in the patient's blood and attack cancer cells. CAR T-cell therapy is used to treat large B-cell lymphoma that has not responded to treatment. CAR T-cell therapy is being studied to treat mantle cell lymphoma that has relapsed or not responded to treatment.
Targeted Therapy for Lymphoma
Targeted therapy is a type of treatment that uses drugs or other substances to identify and attack specific cancer cells. Targeted therapies usually cause less harm to normal cells than chemotherapy or radiation therapy does. Monoclonal antibody therapy, proteasome inhibitor therapy, and BTK inhibitors are types of targeted therapy used to treat adult non-Hodgkin lymphoma.
-
- Monoclonal antibody therapy: Monoclonal antibodies are immune system proteins made in the laboratory to treat many diseases, including cancer. As a cancer treatment, these antibodies can attach to a specific target on cancer cells or other cells that may help cancer cells grow. The antibodies are able to then kill the cancer cells, block their growth, or keep them from spreading. Monoclonal antibodies are given by infusion. They may be used alone or to carry drugs, toxins, or radioactive material directly to cancer cells.
- Proteasome inhibitor drugs work by stopping enzymes from breaking down proteins that are important for controlling cell division.
-
BTK inhibitors for some types of B-cell non-Hodgkin lymphomas work by blocking bruton tyrosine kinase (BTK) protein from helping lymphoma cells multiply.
Radiation Therapy for Lymphoma
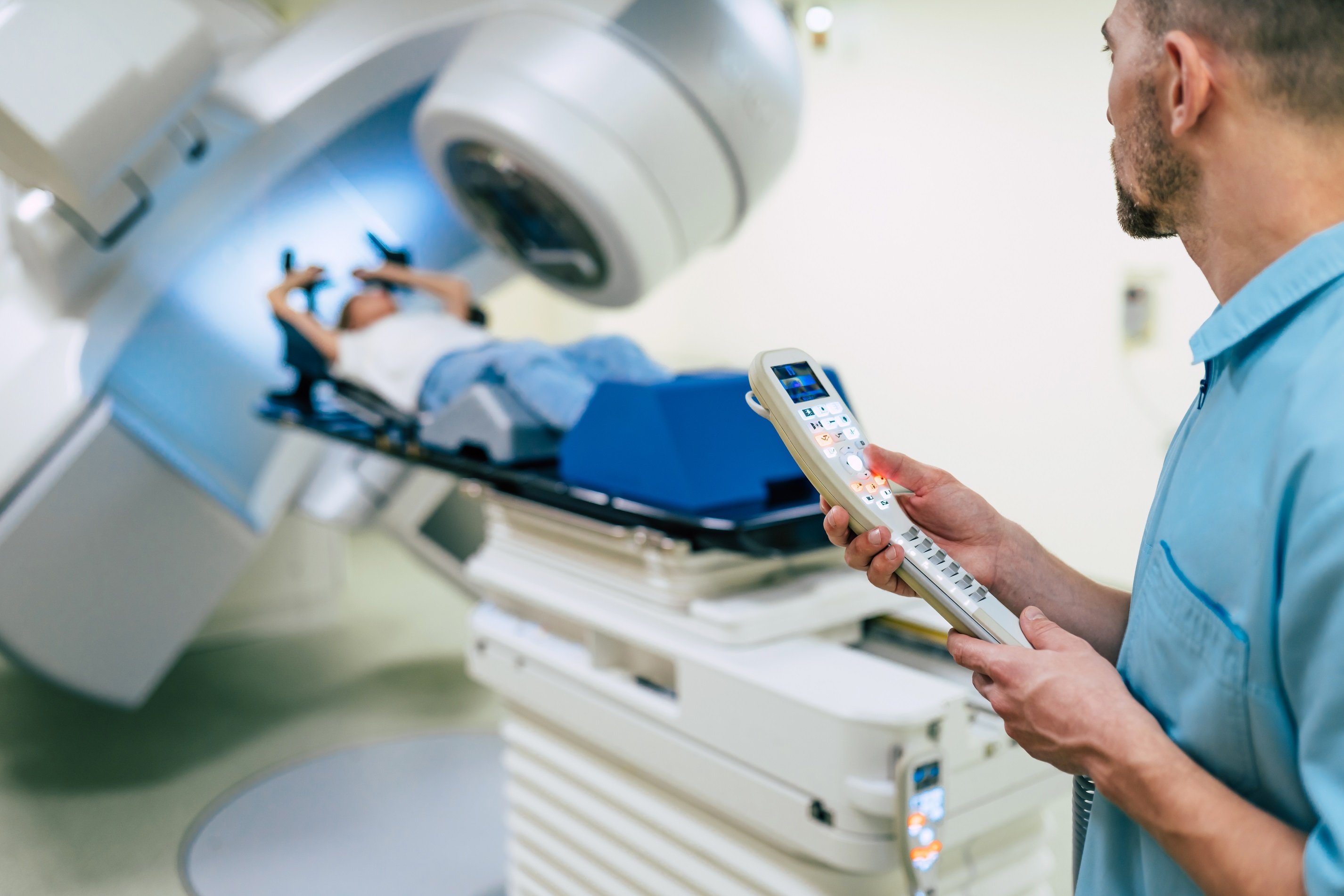
Radiation therapy (also called radiotherapy) uses high-energy rays to kill lymphoma cells. It can shrink tumors and help control pain. Radiation therapy is usually given after or in addition to chemotherapy depending on the non-hodgkin lymphoma subtype.
Two types of radiation therapy are used for people with lymphoma:
- External radiation: This is when a large machine aims the rays at the part of the body where lymphoma cells have collected. Because it affects cells in the treated area only, it is often called local therapy. In most cases, people go to a hospital or clinic for treatment 5 days a week for several weeks.
- Systemic radiation: Some people with lymphoma receive an injection of radioactive material that travels throughout the body. The radioactive material is bound to monoclonal antibodies that seek out lymphoma cells and the radiation destroys the lymphoma cells.
Bone and Blood Marrow Transplantation for Lymphoma
If your lymphoma returns after treatment, you may receive a bone or blood marrow transplantation. A transplant of your own blood-forming stem cells allows you to receive high doses of chemotherapy, radiation therapy, or both. The high doses destroy lymphoma cells as well as healthy blood cells in the bone marrow.
Bone and blood marrow transplants are usually performed in a hospital. Before you receive high-dose treatment, your stem cells are removed, frozen, stored, and in some cases, treated to kill lymphoma cells that may be present. After you receive high-dose treatment to kill lymphoma cells, your stored stem cells are thawed and given back to you through a flexible tube placed in a large vein in your neck or chest area. New blood cells develop from the transplanted stem cells.
The stem cells may come from your own body or from a donor:
- Autologous stem cell transplantation: This is a transplantation in which your own stem cells are used.
- Allogeneic stem cell transplantation: This is a transplantation in which healthy stem cells come from someone other than the person who requires them. Your brother, sister, or parent may be the donor. Or the stem cells may come from an unrelated donor. Doctors use blood tests to be sure the donor’s cells match your cells.
- Syngeneic stem cell transplantation: This type of transplant uses stem cells from a patient’s healthy identical twin.
